|
When these two conditions are met, the equation is said to be balanced. Also the sum of the charges on one side of the equation must be equal to the sum of the charges on the other side. This means that a chemical equation must have the same number of atoms of each element on both side of the equation. Conversions are given between moles, mass, and molecules. Chemical equation: (N H 4) 2 C r 2 O 7 dissociates into C r 2 O 3, N 2, and H 2 O. The Law of Conservation of Mass states that mass is neither created nor destroyed in an ordinary chemical reaction. 4: The Relationships among Moles, Masses, and Formula Units of Compounds in the Balanced Chemical Reaction for the Ammonium Dichromate Volcano. Under the reactant’s side, list Al and O. make a list of all of the elements on each side under the equation for both the reactants and products as shown above. If the elements in a chemical formula are properly capitalized, the smart case converter leaves them as you have typed.Ī balanced chemical equation accurately describes the quantities of reactants and products in chemical reactions. First, identify the elements on the reactant side (left side) and the elements of the compound are on the product side (right side). The equation can be written in lowercase letters.Coefficients are whole number multipliers. Input the chemical formula or compound you want to analyze. When balancing equations, you never change subscripts. Follow these simple steps: Open the oxidation formula calculator. To enter the equation sign, you can use either "=" or "->" or "→" symbols. Add Coefficients To Balance Mass in a Chemical Equation.To enter charge species, just type them as they are, for example Hg2+, Hg22+, or Hg2^2+.Use uppercase for the first character in the element and lowercase for the second. The remaining values will automatically be calculated. The reactants and products, along with their coefficients will appear above. Added by Eddiepow in Chemistry Balances Chemical Equations. Right Hand Side has Sn 1, H 2x2 4, O 1x2 2. All types of parentheses are correct, for example K3 To perform a stoichiometric calculation, enter an equation of a chemical reaction and press the Start button. Again, check the number of atoms in each element.Spaces are irrelevant, for example Cu SO 4 is equal CuSO4.The first reactant set by default by the calculator is $H_ $, $0.Enter an equation of a chemical reaction and click 'Submit' (for example: so32-+cr2o72->cr3++so42- ). The user can enter either the name or the chemical formula for the reactant in this tab. If you are in the habit of inserting any immutable groups in the chemical equation, they must be substituted to avoid any vagueness in chemical compounds. Step 1Īt first, enter the first reactant of the chemical equation in the Reactant 1 window. Our chemical equation balancer developers also suggest that it is wise to remove permanent chemical compounds to avoid any confusion. You can use the Stoichiometry Calculator by following the steps mentioned here. This means that a chemical equation must have the same number of atoms of each element on both side of the equation. In the end, the calculator also provides the user with a wide range of chemical properties of the input reactants and products. It also gives the rate of reaction and the chemical names for the input reactants and products. Following are some equation input format examples: 1. The calculator balances the equation by showing all coefficient proportions. This balancer can also help you check whether the equation is balanced or not, thus you may edit the equation and check its balance. Use the given Periodic Table to add the chemical equation that you need to balance. 
The calculator also outputs the equilibrium constant Kc obtained from the balanced equation. This chemical equation balancer can help you to balance an unbalanced equation. It also provides the chemical structures of reactants and products. Stoichiometry Calculator is an online tool that balances a chemical reaction by equalizing the components of reactants and products resulting in a balanced equation. Stoichiometry defines the relationship between the reactants and products by balancing their components. The chemical reaction also contains some by-products. The reactants chemically react together to give products. It deals with obtaining equilibrium in a chemical reaction to stabilize the reaction. /chemicalequations-58ea60c53df78c516211b81d.jpg) 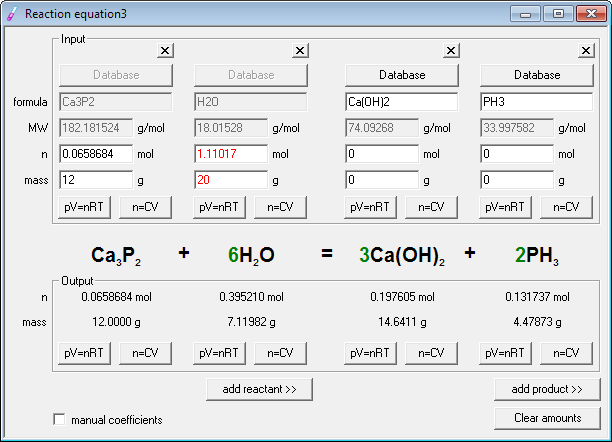
Stoichiometry is an important branch of chemistry that deals with the relationship between quantities. It takes the chemical equation as input and outputs a balanced equation with the equilibrium constant and many other properties of the reactants and products. The Stoichiometry Calculator is used to balance the chemical equations on both sides. Stoichiometry Calculator + Online Solver With Free Steps
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
